Cadmium
Definition
Cadmium is a chemical component and it has the symbol Cd. The soft, bluish-white transition metal is chemically like to the two other metals in group 12, zinc and mercury. Similar to zinc it prefers oxidation state +2 in most of its compounds and similar to mercury it shows a low melting point for a transition metal. Cadmium is a relatively rich element.
Properties
The most common oxidation state of cadmium is +2. Cadmium burn in air to form brown amorphous cadmium oxide .The crystalline form of the same compound is dark red and changes colour when heated, similar to zinc oxide. Hydrochloric acid, sulfuric acid and nitric acid dissolve cadmium by forming cadmium chloride cadmium sulfate.
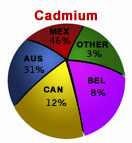
World Resources
Application
About three-quarters of the cadmium used in batteries, mainly in rechargeable nickel-cadmium batteries. Nickel-cadmium cells have a supposed cell potential of 1.2 V. The cell consists of a positive nickel hydroxide electrode and a negative cadmium electrode plate divided by an alkaline electrolyte. More recent nickel-metal hydride batteries reduce the use of Ni-Cd batteries.








